A research team led by Chair Professor Chuxia DENG and Professor Edwin CHEUNG in the Faculty of Health Sciences (FHS) at the University of Macau (UM) has revealed the key molecular mechanism behind resistance to the targeted drug PARP inhibitors in BRCA1-deficient breast cancer, providing a scientific basis for overcoming this type of drug resistance. The team also proposed a related therapeutic strategy to address this resistance. The research has been published in the internationally renowned journal Protein & Cell.
Breast cancer is the most common malignant tumour among women worldwide, and its development is closely linked to genetic factors. About 10% of cases have a familial genetic background, most of which are caused by mutations in the BRCA1 or BRCA2 genes. BRCA1 plays a key role in the process of DNA homologous recombination repair, a repair pathway which is responsible for accurately repairing double-strand breaks and maintaining genomic stability. If this function is impaired, cells rely on other error-prone repair mechanisms, which can lead to genomic instability and cancer development. Based on this defect, PARP inhibitors have been developed as targeted drugs that selectively kill cancer cells with DNA repair defects through a ‘synthetic lethality’ mechanism. They have now become an effective therapy for treating BRCA-mutated breast cancer. However, clinical resistance remains a major challenge that limits their efficacy.
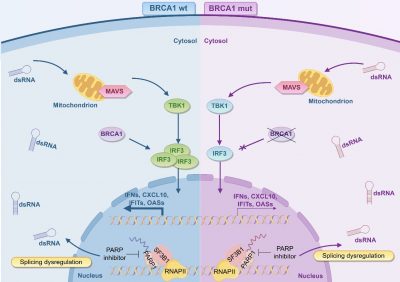
To overcome drug resistance, the research team has revealed a new mechanism through which DNA damage activates innate immune responses. DNA damage not only threatens genomic stability, but also generates cytosolic DNA fragments and abnormal double-stranded RNA (dsRNA). DNA fragments can induce interferon production through the cGAS-STING pathway, thereby enhancing anti-tumour immune responses. Meanwhile, dsRNA triggers MAVS-mediated anti-viral mimicry innate immune responses. These findings open new avenues for combination therapy involving the use of STING agonists alongside PARP inhibitors. Although the detailed mechanisms of the dsRNA-MAVS pathway are yet to be elucidated, the combination of targeted DNA repair and the activation of immune responses shows promise in overcoming resistance and improving breast cancer treatment outcomes.
Through functional proteomics, the research team discovered that PARP inhibitors can enhance the interaction between PARP1 and the splicing factor SF3B1. This disrupts spliceosome function, leading to the accumulation of dsRNA and the activation of an anti-viral mimicry innate immune response. The study also revealed a new function of BRCA1 in regulating innate immunity. BRCA1 deficiency leads to the downregulation of IRF3 expression, thereby weakening this immune response and reducing the sensitivity to PARP inhibitors, resulting in intrinsic resistance. Notably, the dsRNA analogue poly(I:C) can effectively activate this immune pathway, and combining it with PARP inhibitors significantly enhances their killing effect on BRCA1-deficient breast cancer cells. Animal experiments confirmed that this combination strategy can significantly improve the tumour suppression rate. This study elucidates a new mechanism by which BRCA1 deficiency leads to resistance by downregulating the IRF3-mediated innate immune signalling pathway. It also puts forward the innovative approach of reversing this resistance by activating the pathway with poly(I:C), providing a new strategy for overcoming PARP inhibitor resistance in BRCA1-deficient breast cancer.
The corresponding authors of this study are Prof Deng and Prof Cheung, with Cuiting ZHANG, PhD candidate in UM FHS, as the first author. Other team members include Associate Professor Terence, Chuen Wai POON, Adjunct Associate Professor Xiaoling XU, Assistant Professor Kai MIAO, and Postdoctoral fellows Jingbo AHOU and Josh, Haipeng LEI in UM FHS. The study was supported by the Natural Science Foundation of China (File No.: 82030094), the Science and Technology Development Fund of the Macao SAR (File Nos.: 0009/2022/AKP, 0054/2023/RIA1, 0129/2024/RIA2, and 0137/2020/A3), and the University of Macau (File No.: MYRG-GRG2024-00073-FHS). The full version of the research article is available at: https://academic.oup.com/proteincell/advance-article/doi/10.1093/procel/pwaf104/8419885?login=false.