Cardiac muscles cells are considered difficult to regrow and the differentiation process is costly. To address this problem, a research team from the Faculty of Health Sciences (FHS), University of Macau (UM), invented two new methods to generate heart cells in vitro with high consistency and quality, without using expensive and complicated supplements. This is a key step toward the generation of heart cells for cell therapy and drug screening. The study has been published in the renowned journal Cell Reports.
Titled ‘Endogenous Insulin-like Growth Factor (IGF) Signaling Directs Heterogeneous Mesoderm Differentiation in Human Embryonic Stem Cells’, the study was conducted by a team led by Associate Professor Guokai CHEN from the FHS, along with his PhD students Yang YANG and Zhili REN. Chair Professor Wei GE, Assistant Professor Qi ZHAO, Senior Instructor Weiwei LIU, as well as researchers from Nanjing also participated in the project. IGF is well-known for its role in stem cell survival and bone growth, but its role in cell fate determination was not clear. Prof Chen’s team has found that the IGF signal influences the potential to generate different mesodermal and endodermal cell types from human embryonic stem cells (hESCs). During the early stages of hESC differentiation, IGF promotes liver and endothelial cell types, but prevents cardiac differentiation. The delicate balance of IGF signal is critical for various cell types to emerge simultaneously during embryogenesis. When the endogenous IGF pathway is suppressed, heart cells are induced from hESCs.
At present, WNT inhibition is a common modulation used in vitro to generate heart cells. However, the regulation is much more complicated in vivo during embryogenesis. This study reveals an important regulatory mechanism in heart cell differentiation, which leads to the development of two new methods to produce heart cells, through IGF pathway inhibition and CK2 kinase inhibition.
The findings from this study provide alternative ways for the in vitro production of cardiomyocytes, improve our understanding of cell fate diversity in early embryogenesis, and allow researchers to guide cell fate specification towards target cell types based on the new knowledge. This project was jointly funded by UM and the Macao Science and Technology Development Fund. A patent application for the technology has been filed. For more information, please visit
https://www.sciencedirect.com/science/article/pii/S2211124719315293.
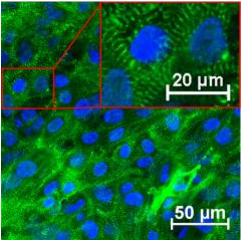
Cardiac muscle cells differentiated in vitro from human embryonic stem cells

The research team

