A team led by Tzu-Ming LIU, Associate Professor in the University of Macau (UM) Faculty of Health Sciences (FHS), has made a breakthrough in the development of label-free anti-cancer drug screening method. They found lipofuscin-like red autofluorescence could early predict the fate of cell death, and time-course monitor the treatment responses in 3D tumour-culture models. Also, this endogenous fluorescent marker could visualize the apoptosis in live zebrafish embryos during development. Moreover, the fluorescence lifetime analysis of lipofuscin-like red autofluorescence can further distinguish cell necrosis from apoptosis. Such lipofuscin-like red autofluorescence hallmark of cell death can extend the information dimension of pharmacodynamics for organoid research and application. The study has received considerable attention in the field of biomedicine and has been published on the Redox Biology.
Apoptosis is a frequently occurring and tightly regulates biochemical process for the maintenance of tissue homeostasis. Dysregulation of apoptosis may lead to uncontrolled proliferation and malignant tumours. Cancer treatment relies on therapeutic compounds or cell drugs that trigger the apoptosis pathway. With more understanding of cell death mechanisms, several biochemical features have been exploited to detect dying and dead cells. However, these approaches require fixation and staining of tissues, which limits the possibility of time-course evaluation. To visualize the pharmacodynamics and the apoptosis response, scientists have designed live-cell staining probes. All these reporter assays relied on exogenous reagents, synthetic nanoprobes, or complicated transfection protocols, which may alter the physiology of cells under test. Alternatively, endogenous fluorophores such as NADH and flavins in cells may enable quick and label-free evaluation of cell senescence. However, these metabolic fluorophores are nonspecific to the cell senescence process and are easily affected by other metabolic activities. Therefore, the development of a detection method that uses endogenous fluorophores and can specifically identify cell-death processes is of great significance for revealing pharmacodynamics and testing drug sensitivity.
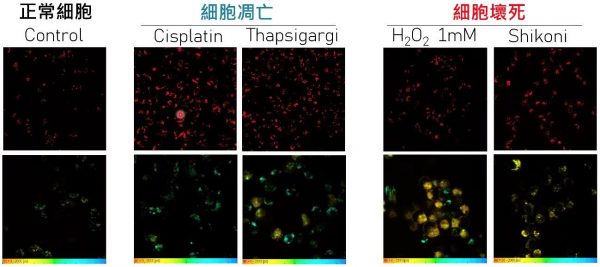
Lipofuscin is a brown-yellow, autofluorescent pigmented granules that accumulates in cells during aging. In the past, lipofuscin has been widely used to report the degeneration of the retina, neurons, cardiac muscles, and human mesenchymal stromal cells. Prof. Liu’s research team used two-photon fluorescence microscopy and spectroscopy to observe the intensity of lipofuscin-like red autofluorescence. The fluorescence intensity of lysosomal lipofuscin in apoptotic and necrotic cells was significantly increased, compared with the normal cells. Meanwhile, stress-induced autofluorescence lifetime can further distinguish necrosis from apoptosis. The team then successfully applied this label-free cell death detection method on avatar drug testing system in cancer precision medicine, including spheroids, patient-derived organoids (PDOs) and three-dimensional tumour slice culture (3D-TSC) model which contains immune cells. Through continuous pharmacodynamic monitoring, this technique can optimize drug susceptibility assessment, overcome individual pharmacodynamic differences due to different genetic backgrounds and tissue heterogeneity, and improve the throughput of drug screening.
Prof. Tzu-Ming LIU, FHS Dean Chuxia DENG, Professor Kathy Qian LUO and Associate Professor Leo Tsz On LEE are the corresponding authors of the study, and PhD students, Yinghan YAN and Fuqiang XING, share the first authorship. Others who have contributed to the study include FHS Research Assistant Jiayue CAO, PhD student Yiling HU, etc. The project was supported by the Science and Technology Development Fund, Macao SAR (file number: 122/2016/A3, 018/2017/A1, 0011/2019/AKP, 0002/2021/AKP; 0007/2021/AKP, 0120/2020/A3, 0026/2021/A) and UM (file number: MYRG2018-00070-FHS). The full version of the research article can be accessed at https://www.sciencedirect.com/science/article/pii/S2213231722003500?via%3Dihub.