A research team led by Joong Sup SHIM, Associate Professor in the Faculty of Health Sciences (FHS) at the University of Macau (UM), in collaboration with researchers from Chongqing Medical University and Renji Hospital of Shanghai Jiaotong University, has discovered an unrecognised therapeutic vulnerability in VHL‑deficient renal cell carcinoma (RCC), the most common form of kidney cancer. The study shows that RCC cells lacking the tumour suppressor von Hippel-Lindau (VHL) are highly sensitive to DNA methyltransferase (DNMT) inhibitors, revealing a promising precision medicine strategy for the major RCC subtype. The findings have been published in the international journal Experimental & Molecular Medicine.
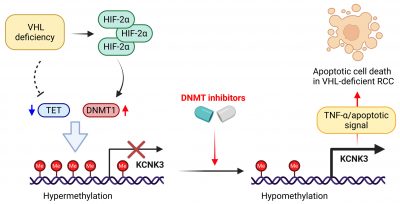
Renal cell carcinoma (RCC) is often caused by the loss of the VHL gene, which stabilises HIF‑2α and alters cellular metabolism and angiogenesis. Recent studies have also suggested that VHL inactivation triggers widespread DNA hypermethylation. In order to leverage this epigenetic state to enhance therapeutic efficiency, the research team performed a synthetic lethal drug screen using VHL‑isogenic RCC models. The results showed that FDA‑approved DNMT inhibitors, such as decitabine and azacitidine, as well as the investigational agents RX‑3117 and SGI‑1027, selectively suppress the growth of VHL‑deficient RCC cells.
Mechanistic studies revealed that VHL deficiency results in HIF‑2α-driven upregulation of DNMT1, which leads to widespread CpG hypermethylation. Among the genes silenced by this epigenetic remodelling, the team identified KCNK3, a potassium‑channel tumour suppressor, as a key mediator of synthetic lethality induced by DNMT inhibitors. The KCNK3 promoter is hypermethylated and repressed in VHL‑deficient RCC, but DNMT inhibitors can reverse this methylation, thereby restoring KCNK3 expression and triggering apoptotic signalling.
In mouse xenograft models, decitabine was found to selectively inhibit the growth of VHL‑deficient RCC tumours. However, this effect was largely abolished when KCNK3 was depleted, demonstrating strong therapeutic specificity. The team further validated this finding using patient‑derived RCC cell models, in which tumour cells harbouring pathogenic VHL mutations exhibited markedly higher sensitivity to DNMT inhibitors than VHL‑wildtype samples.
This study provides compelling evidence that DNMT inhibition represents a synthetic lethal strategy for VHL‑deficient RCC, offering a new therapeutic avenue for patients with this common genetic alteration. Through the integration of synthetic lethal screening, epigenetic profiling, mechanistic dissection, and patient‑derived models, the research establishes a strong foundation for future clinical translation.
The corresponding authors of the study are Prof Shim, Yongjun DANG, Dean of the School of Pharmacy at Chongqing Medical University, and Jianfeng WANG, Chief Physician at Renji Hospital of Shanghai Jiao Tong University. The first authors are Yue PU, a PhD Candidate at UM, and Ziruoyu WANG from Chongqing Medical University. Other contributors to the research include Associate Professor Gang LI, Assistant Professor Kai MIAO, and Senior Instructor Kaeling TAN in UM FHS. This project was funded by the Science and Technology Development Fund of the Macao SAR (Grant Nos.: 0139/2024/AFJ and 0049/2022/A), the Shenzhen Science and Technology Innovation Commission (Grant No.: EF2023‑00070‑FHS), the University of Macau Development Foundation and the University of Macau (Grant Nos.: MYRG‑GRG2023‑00124‑FHS‑UMDF, MYRG‑GRG2024‑00056‑FHS, and MYRG‑GRG2025‑00182‑FHS), Joint Funding Scheme for NSFC‑FDCT Project (Grant No.: 22461160267), and the Ministry of Education Frontiers Science Center for Precision Oncology, University of Macau (Grant No.: SP2023-00001-FSCPO). The full version of the research article is available at: https://www.nature.com/articles/s12276-026-01663-w.