A team led by Qi ZHAO, an associate professor of the Faculty of Health Sciences (FHS) at the University of Macau (UM), has made important progress in the development of nanomedicines targeting triple-negative breast cancer (TNBC). They successfully prepared NIR-II light excitation nanomedicines through a metal ion-mediated coordination self-assembly strategy, which can achieve NIR-II phototheranostics towards TNBC through aptamer-mediated active targeting, simultaneously amplifying the therapeutic efficiency of copper ion induced oxidative stress and cuproptosis, enchance the therapeutic efficiency. The research has been published in Chemical Engineering Journal.
Chemotherapy is currently still main treatment modality of TNBC in clinical. Unfortunately, it is failed to improve life quality of breast cancer patients by systemic administration due to severe side effects induced by non-specific drug biodistribution. The phototheranostics in the second near-infrared window (NIR-II, 1000-1700 nm), have shown great prospects in breast cancer theranostics, ascribed to deeper tissue penetration and higher maximum permissible exposure to lasers resulted from the depressed photon absorption and scattering by biological samples.
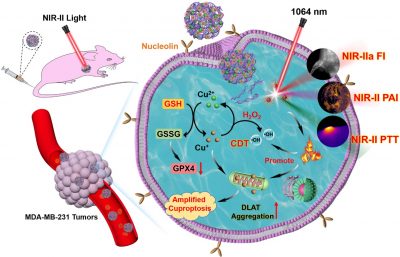
Cuproptosis is a copper induced cell death pathway occurring in the tricarboxylic acid (TCA) cycle, involving in mitochondrial lipoylated protein aggregation accompanied by the loss of iron-sulfur (Fe-S) cluster proteins. The binding of lipoylated components and copper will cause high proteotoxic stress and eventual cell death. Furthermore, it has been widely reported that copper ions can severe an oxidative stress inducer for chemodynamic therapy (CDT) through Fenton-like reaction induced hydroxyl radicals (·OH) generation in tumour tissues. However, intracellular glutathione (GSH) mediated copper-chelation, the easy copper efflux and the lack of targeted copper delivery may attenuate Cu-based therapeutic efficacy.
Herein, the research team developed a NIR-II excitation multifunctional nanomedicine by self-assembly of targeting aptamers (AS1411) utilises metallic copper ions as coordination nodes, followed by coated with ultrasmall NIR-II polymer dots (Pdots) with excellent optical performances, forming a carrier-free hybrid nanosystem. After positive targeting into TNBC through specific recognition of aptamer towards cell surface nucleolin, Pdots loaded in nanomedicine enable NIR-IIa fluorescence/NIR-II photoacoustic imaging-guided NIR-II photothermal therapy (PTT) under NIR-II (1064 nm) light excitation, achieving precise phototheranostics of TNBC tumours. Overexpressed GSH and high level of ferredoxin 1 (FDX1) reduce Cu(II) to cytotoxic Cu(I) in tumour cells. Cu(I) can not only catalyse endogenous hydrogen peroxide (H2O2) to generate ·OH for efficient CDT, but also bind to lipoylated components within mitochondria, leading to abnormal aggregation of dihydrolipoamide S-acetyltransferase (DLAT) and thus inducing cuproptosis through proteotoxic stress. The NIR-II photothermal effect and GSH depletion can amplify the therapeutic effect of copper-mediated oxidative stress and cuproptosis, thus augmenting therapeutic efficiency towards TNBC. This work proposes a promising strategy for targeting multimodal theranostics towards TNBC.
Qi ZHAO is the corresponding author of the study, and UM’s postdoctoral fellow Yeneng DAI is the first author. Others who have contributed to the study include FHS PhD students Dongliang LEONG, Ziang GUO, Jiaqi WANG, Yuheng GU and Yingjun PENG, and Postdoctoral Fellow Lipeng ZHU. The project was supported by the Science and Technology Development Fund of Macau (File nos. FDCT/0043/2021/A1 and FDCT/0002/2021/AKP), the National Key R&D Program of China (2019YFA0904400), and the UM (File no. MYRG2022-00143-FHS).